Dig, dig, dig, bite! Dig, dig, dig, bite!
Like an inmate who chips away at the same square foot of floorboard day after day with the hope of tunneling to freedom, Otis returns to the same rug to dig day after day. The most obvious motivation for attempting to paw his way through to the unit below ours would be a uniquely delicious smell housed deep in the recesses of this particular rug’s carpet fibers. But Otis is an equal opportunity digger, and enjoys digging into another rug at his grandparents’ house whenever he goes for a visit.
When viewed from above with his white underbelly and brown overcoat, Otis blends into the surface of the rug like a great white shark blends into the murky grey of a tumultuous ocean. Otis’s rate of digging progressively accelerates like a careless archeologist who has glimpsed part of a long-searched for relic. At some point the frustration over a lack of progress overcomes Otis and he sinks his teeth into the rug, huffing and puffing with a mouthful of carpet as he catches his breath. Once Otis feels like the rug has learned its lesson, he relinquishes his toothy grasp and repeats the digging-biting process all over again.
Not only does Otis return day after day to the same rug, but he also digs on the exact same spot on this unfortunate tapestry. The spot in question contains an image of a green and brown flower, the largest of a repeating flower pattern, situated in the center of our entryway rug. Additionally, the rug that Otis is fond of digging on at his grandparent’s house contains a different floral pattern, but again Otis locates the biggest and most central flower before commencing his drilling operation.
Otis has decreased his archeological digs thanks to our preservational training efforts that aimed at maintaining the historical integrity of our floor adornments. That being said, Otis was never trying to rip or penetrate the spot he was digging, he was content simply digging without goal or expectant result. The reader should also note that Otis turns his nose up at digging into actual soil or flowerbeds (a pretension that his grandparents are quite thankful for).
Explanations for Otis’s digging have ranged from delving into the history of the French Bulldog breed to ascribing an anthropomorphized desire to rid the world of hideous rugs. French Bulldogs are thought to have stemmed from breeding miniature English Bulldogs with terriers and/or pugs. If terriers did indeed contribute to Otis’s gene pool, then the instinctual digging could at least be partially explained.
And yet, the question of what exactly Otis is thinking when he attempts to remove the pesky flower from its threadbare garden will likely never be answered. But the unanswerable question leads to a more general, and perhaps more answerable one: Can Otis think? And if he can think, then to what capacity?
In today’s article we will not limit ourselves to the investigation of my neurotic French Bulldog. Instead, we will consider the larger issue of nonhuman animal consciousness.
Despite our high-minded sensibilities, we humans are still animals. However, instead of wasting digital ink by repeating “nonhuman” before every reference to animals, I will refer to nonhuman animals as simply “animals” from this point forward.
Before we begin our discussion of what animal consciousness is we must be clear about what it is not. There is a strong urge to anthropomorphize animal consciousness by projecting human thinking onto the animal mind. We must be careful about this very natural desire to extend the moral, logical, and conceptual underpinnings of our own conscious experience to animal consciousness.
Humans evolved as social creatures that developed moral codes as a way of sustaining the social order. The role of a clawless, relatively toothless, and comparatively physically vulnerable bipedal ape drove the evolving human brain to emphasize computational power and mental abstraction over grit and brawn. These forces colluded to create the experience of human consciousness. The same evolutionary forces do not apply to dogs, rabbits, or mice. Each animal possesses a brain, and likely a consciousness, that reflects the animal’s position in the specific ecological niche that evolutionary forces carved out for it.
We will examine a large amount of experimental evidence to ensure that we have as many data points regarding animal consciousness as possible. The evidence will not point to a universal experience of a humanoid consciousness; instead it will emphasize the unique corner of experiential consciousness that a given animal occupies. Animals need not think like us to think.
To begin to answer the question of animal consciousness we must first decide what we mean by “consciousness.” Due to the immense complexity of the concept of consciousness, there are a near infinite number of definitions that span the gambit of academic departments from philosophy to neuroscience.
Perhaps most simply, consciousness has been defined as what it is like to be something (Squire, 2013). In other words, consciousness defines the experience of what it is like to wake up in the morning, enjoy a cup of coffee, and perceive the sun streaming in through your window.
For our purposes, we will have to go beyond a vague philosophical definition of consciousness to a more precise scientific definition. To accomplish this feat I will stand on the shoulders of the biologist Gerald Edelman and his concept of primary and secondary consciousness. Primary consciousness has also been referred to as sensory consciousness, and it is this latter nomenclature that I will utilize in our ensuing discussion.
Sensory consciousness describes the subjective perception of sensory information. In other words, sensory consciousness refers to the subjective experience of the green color of a leaf, the smell of Thanksgiving dinner, or the emotional quality of fear. (Edelman, 2003)
Sensory consciousness can be contrasted with secondary consciousness, or higher-order consciousness, as it is also known. Higher-order consciousness refers to our ability to plan for the future and mentally revisit the distant past. Abstraction, metacognition (“knowing about knowing”), and self-reflection are all examples of higher-order consciousness. (Edelman, 2004)
For the purpose of today’s article, we will be primarily concerned with whether or not animals possess sensory consciousness. Thus, when I refer to “consciousness” from this point on I will be referring to sensory consciousness. A great deal of evidence that we will discuss today suggests that higher-order consciousness may not be unique to humans. But in order to avoid a philosophical quagmire, I will limit the scope of my discussion to sensory consciousness and allow the reader to draw his or her own conclusions regarding higher-order consciousness in animals.
How does human consciousness arise?
There are many hypotheses as to how the brain generates consciousness. We will examine one of the front-runners, but please keep in mind that the complete understanding of the neurobiological underpinnings of consciousness lies somewhere far beyond the current scientific horizon.
Let us begin by muddying the waters a bit with a discussion of free will. The notion that the Self that experiences our consciousness freely and willfully chooses all of our behavior is fundamental to our understanding of our internal world. And yet, when we look closely at the neuroscience of free will, we begin to see that the issue of agency is a great deal more complex than it first appears.
Multiple experiments have examined the relationship between brain activity and the subjective experience of free will. The typical free will experiment involves using electroencephalographic (EEG) or functional magnetic resonance imaging (fMRI) technology to monitor the activity of participants’ brains while they perform a willful action (randomly snapping their fingers, giving a thumbs up, etc.). Experiment after experiment has shown that the brain engages motor units seconds before the participant has the experience of willing his or her action. Thus, free will may be better understood as a sensation constructed after a behavior is enacted, rather than an executive order that triggers an action. (Hallett, 2007)
So does this mean that we are simple robots without free will?

MOTOR CORTEX “Human motor cortex” by Cortex sensorimoteur1.jpg: Pancratderivative work: Iamozy – Own work, This file was derived from: Cortex sensorimoteur1.jpg: . Licensed under CC BY-SA 3.0 via Wikimedia Commons.
No. The brain is a highly redundant circuit and any one part is interconnected either directly or indirectly with the whole. Thus, the activation of the motor cortex that triggers our actions can be thought of as representing the entirety of all brain input at any one time: the summative brain state. Thus, our brains do decide what to do; it just may be that the initiation of action is quite a bit less conscious than we typically imagine it to be.
Free will may better be characterized as free won’t in that our prefrontal cortex doesn’t seem to initiate movement, but instead holds veto power over its final execution. And the actual experience of free will may, in actuality, be the result of the performed action lining up with the expected action. In other words, our brains hunger for causality, so if an action lines up with a thought, then the brain reasons that the thought must have caused the action. (Hallett, 2007)
Obviously free will is a complex topic and could easily be expanded into an entire article. The reason we discuss it here is to begin to close the perceptual gap between human and animal consciousness. The argument that animals lack consciousness based on their sometimes-stereotyped behavior and relative lack of free will falls apart when we look closely at the illusion of our own free will.
Regardless, of the reality or unreality of free will, how is our experience of consciousness generated?
The brain is a highly recurrent circuit that constantly reviews and refines information. In general, sensory information gathered from our sense organs (ears, eyes, nose, mouth, and touch-sensitive nerve bundles in our skin) proceeds from the back of our brain towards the front, increasing in perceptual complexity along the way. For example, a single visual percept that begins at the back of the brain is combined with multiple visual, auditory, and somatic perceptions along its path to the front of the brain. (Squire, 2013)
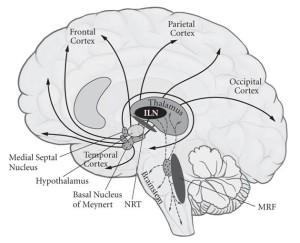
THALAMUS “NeuralCorrelatesOfConsciousness2” by Original uploader was Fmorm at en.wikipedia – Transferred from en.wikipedia. Licensed under CC BY-SA 3.0 via Wikimedia Commons.
The outer portion of our brain is known as the neocortex and is responsible for much of the refining process that generates progressively more elaborate sensory representations of our external environment. The neocortex is intimately connected to the thalamus/“Relay,” a structure that sits at the epicenter of the brain and is responsible for relaying sensory and motor information from the body to the brain.
The thalamus/“Relay’s” role turns out to be more complex than acting as a simple output for relayed information from the body to the cortex. The thalamus/”Relay” also receives input from the cortex that is capable of amplifying or decreasing specific incoming sensory or motor signals. In this way, the thalamus/”Relay” can increase the “volume” of sensory information of interest and filter out information that is of less immediate interest.
Furthermore, the input and output signals between the thalamus/”Relay” and cortex set up a resonance circuit that generates a cortically-comprehensive sensory experience. The dynamic core theory of consciousness posits that the elaboration of sensory information courtesy of the thalamo-cortico-thalamic network produces the state of sensory information integration that we experience as consciousness.
Thus, consciousness is suggested to be an emergent property of a highly complex system of integrated sensory information. There are many theories that elaborate on the dynamic core theory (the current leader seems to be the informational integration theory of consciousness), but they are beyond the scope of this article. (Tononi & Edelman, 1998)
So if consciousness is an emergent property of any highly integrated sensory system, what leads us to believe that the equally complex animal brain cannot experience consciousness? In short: nothing.
In the 1600s Descartes famously wrote, “I think, therefore I am,” in his work Discourse on the Method. Descartes went on to write a great deal of highly influential philosophical commentary, but one in particular is of concern when considering the history of animal consciousness. Descartes argued that animals lacked a soul, consciousness, and could not experience pain. Descartes believed that animals were mechanistic automata that operated purely at an instinctual level. It may be difficult to believe, but this view of animal consciousness persisted for centuries afterward in the scientific mainstream.
There were glimmers throughout history of scientists who did not take Descartes conclusions regarding animal consciousness for granted. Darwin argued in his Origin of Species that the closer one looks at animal behavior the more difficult it becomes to simply ascribe instinctual causation for their behaviors. (Darwin, 2009)
In the heyday of behavioral psychology during the early to mid 1900s, the two leading behaviorists Watson (1) and Skinner (2) roundly rejected the notion that animal behavior should be explained by anything beyond instinctual mechanisms. ((1) Watson, 1913; (2) Skinner, 1977)
It wasn’t until the 1970s that the zoologist Donald Griffin argued that animal consciousness needed to be studied more empirically. Griffin claimed that the behavioral flexibility and adaptability of animals to novel situations should be viewed as evidence of animal consciousness. (Griffin, 2001)
Finally in 2012 a group of neuroscientists signed the Cambridge Declaration on Consciousness, unequivocally asserting that animals experience consciousness (Low et al., 2012). The group cited an overwhelming body of evidence supporting the presence of consciousness in many species of animals. Anyone who has owned a pet or observed an animal for any duration may take the aforementioned declaration as a rather redundant restatement of the obvious. But as we saw in the preceding paragraphs, the scientific community has been battling over the issue of animal consciousness for centuries. It is with this battle in mind that we will start at the most basic of conscious experiences and build from there.
Rather than try to definitively “prove” that animals experience consciousness, I will take a tact utilized by Griffin and Speck in their 2004 article entitled New Evidence of Animal Consciousness. Griffin and Speck suggested that instead of certainty, we look for probability. If, as many have suggested, it is “impossible” to know whether animals experience consciousness, then we must assert that there is a 50% chance that animals are conscious and a 50% chance that they are not (Griffin & Speck, 2004). Thus, from a starting point of a coin toss, experiments will either push our certainty of animal consciousness towards 99% or towards 1%. So instead of arguing over 100% or 0% certainty, my article will make the case that there is a high degree of probability that animals experience consciousness.
To establish this high degree of probability we will look at various components that make up the subjective experience of human consciousness and ask what science has to tell us about the animal experience of these qualities of consciousness.
First and foremost, let’s return to the neurobiological underpinnings of human consciousness and ask what structural differences in the brains of animals lead us to believe that they are not capable of consciousness.

EVOLUTION “Human evolution scheme” by M. Garde – Self work (Original by: José-Manuel Benitos). Licensed under CC BY-SA 3.0 via Wikimedia Commons.
As of today, scientists have yet to identify any unique structure in the human brain that is responsible for the conscious experience (Squire, 2013). In fact, there is remarkable continuity in the structure of the mammalian brain. The first mammal is thought to have diverged 220 million years ago (mya), primates around 85 mya, and Homo sapiens (the direct ancestor of modern man) only 400,000 years ago (Cavalli-Sforza & Feldman, 2003). Thus, the structural overlap between our brains and the brains of animals should come as no surprise considering the relatively inconsequential amount of evolutionary time that modern humans have enjoyed their unique evolutionary path. Thus, from a neuroanatomical standpoint, the dynamic core theory with its recurrent thalamo-cortico-thalamic circuits should apply equally to humans and animals.
It might not be too hard to buy into an ape experiencing consciousness as a result of its similar neuroanatomical architecture, but is this overlap in neuroanatomical structure also true of the brains of animals like rats that individuated around 20 mya (Kay & Hoekstra, 2008)? As it turns out the answer is yes and is the reason that so much of our knowledge of our own neurological mechanisms have been extrapolated from experiments with rats (Griffin & Speck, 2004).
So to sum up, there is no neurobiological reason to expect that animals do not experience consciousness. (Griffin & Speck, 2004)
From the basic neurological underpinnings of consciousness let us now turn to the basic conscious experience of a visual perception. The ability to consciously experience visual imagery is fundamentally different from, for example, the simple reflexive reorientation of one’s head to a quickly moving object. It may seem ridiculously elementary to ask if an animal is conscious of what it sees, but as we saw from our brief historical review, the topic of animal consciousness is contentious to say the least. And in building a structure of evidence in support of animal consciousness we must lay a strong and basic foundation. The foundational question of whether animals are conscious of visual images can be answered by studying a phenomenon known as blindsight.
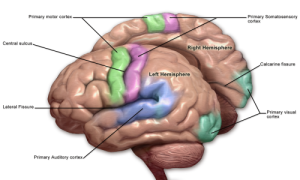
VISUAL CORTEX “Blausen 0103 Brain Sensory&Motor” by BruceBlaus. When using this image in external sources it can be cited as:Blausen.com staff. “Blausen gallery 2014“. Wikiversity Journal of Medicine. DOI:10.15347/wjm/2014.010. ISSN 20018762. – Own work. Licensed under CC BY 3.0 via Wikimedia Commons.
Blindsight is a phenomenon in which damage to the visual cortex (V1) produces an area of conscious blindness in a section of the visual field in spite of a somewhat intact, albeit unconscious, visual system for that blind spot. Visual information normally travels from the retina of the eye along the optic nerve to the superior colliculus and the lateral geniculate nucleus (LGN) in the thalamus/“Relay.” From the LGN, visual information is sent to V1 and from there to many further visual association areas throughout the neocortex. V1 seems to play a central role in the conscious experience of vision because damage to this structure eliminates our conscious experience of vision (blindsight) while preserving some of the more basic visual abilities. (Squire, 2013)
Let’s look at an example to understand what I mean. Human patients suffering from blindsight will deny any perception of a glass of water placed in their blind spot in spite of their ability to reach out and precisely grasp it. In other words, despite a conscious blindness to a glass, patients can extend their arms, adjust the rotation of their hands, and open and close their hands precisely around a glass that for all purposes doesn’t “exist” for them consciously. Additionally, patients suffering from blindsight can report a “sense” of motion as an object moves through their visual field despite a complete lack of conscious experience of having viewed the object.
Many theories that seek to explain blindsight point to evolutionarily ancient structures such as the superior colliculus that generate the unconscious responses to visual stimuli. No matter the explanation, the phenomenon of blindsight provides a way for scientists to measure the conscious experience of visual perceptions.
Thus, if an animal were capable of experiencing blindsight, then we would have to conclude that they are capable of consciously experiencing visual percepts. As it turns out, well-crafted experiments have repeatedly demonstrated blindsight in monkeys (Stoerig, Zontanou, & Cowey, 2002).
Monkeys with V1 lesions that created a blind spot in their visual field were trained to press a square (visible in the preserved section of their visual field) on a computer screen to receive a food reward. After a great deal of training the monkeys could correctly identify and press the square for a reward even when it fell into their blind spot. The monkeys were then trained to press another button to indicate the absence of the square when it was absent from the screen. The surprise came when these same monkeys were again presented with the original food related square in their blind spot. Consciously blind monkeys who had previously pressed the square for food despite it falling in their blind spot no longer touched the square; instead they pressed the button to indicate an absent conscious experience of the square.
Other experimental designs have reaffirmed the conclusion of the aforementioned experiment: monkeys do indeed experience blindsight and thus, consciously experience visual images. This is a small but important brick in the foundation of a conceptual transition from animals as simple reflexive automatons to conscious organisms. Of note, the lack of evidence of blindsight in other animals does not necessarily reflect their lack of a conscious experience of visual perceptions, but instead is due to technical limitations of percept-reporting capability in non-primate animals.
So there seems to be evidence that animals are conscious of the images they view, what other components of consciousness have been investigated?
A seemingly vital component of human day-to-day consciousness is the experience of memory. Not only can we re-experience past events courtesy of our memory, but we can also use this historical information to refine our current actions. Many mammals from squirrels (1) to dolphins (2) demonstrate various degrees of memory ability ((1) Templer & Hampton, 2013; (2) Griffin & Speck, 2004). But perhaps the most surprising evidence of memory and cognition in animals comes from birds.
Why is it so surprising that birds have the ability to remember and utilize said memory in complex cognitive models? Because birds lack the neocortex that was so critical to our discussion of human consciousness (e.g. thalamo-cortico-thalamic recurrency). Granted, birds do have their own rough equivalent of the neocortex known as the dorsal ventricular ridge, but, even so, the fact that birds demonstrate evidence of consciousness is remarkable in that it suggests that the phenomenon of consciousness evolved separately (or perhaps once in our shared ancestors). This would suggest that consciousness is an adaptive trait that was selected for because of the advantage it provided an organism. (Clayton & Dickinson, 1998)

SCRUB JAY “Western Scrub Jay” by Ingrid Taylar from San Francisco Bay Area – California, USA – Western Scrub Jay. Licensed under CC BY 2.0 via Wikimedia Commons.
Let’s look to a member of the corvid family, the scrub jay, to learn more about how birds remember and what they do with their memories. Corvids include such other well-studied birds as the crow and the raven.
Scrubs jays are capable of learning that specific foods spoil after a particular period of time. Scrub jays are then able to compare the memory of when they stored the food with the amount of time that has passed (as long as 5 days!) to determine edibility. If a stored food reserve is past its “expiration date,” the scrub jay will abandon the stash. (Clayton & Dickinson, 1998)
Even more remarkable, scrub jays that steal from their neighbor’s food stash demonstrate an interesting version of metacognition (thinking about what others think). The kleptomaniacal scrub jays will re-hide their own food only if another scrub jay (innocent) saw them hide it. This observation is remarkable because it suggests that the filching scrub jay thinks other scrub jays burglarize as well. The criminal scrub jay then attempts to avoid being robbed by relocating the food stash, but only if another scrub jay (albeit innocent) has watched the looter hide its food. (Emery & Clayton, 2001)
From blind monkeys to pilfering birds, we have already plotted quite the investigational course. Let’s turn to the issue of self-recognition.

MIRROR TEST “Mirror test with a Baboon” by Moshe Blank – Own work. Licensed under CC BY-SA 3.0 via Wikimedia Commons.
I take for granted that the man in the mirror whose hair I comb every morning is not only complicit in the act, but represents my own image. The recognition that a reflected image is oneself turns out to be a fairly complex form of consciousness. The so-called “mirror test,” operationalized by the psychologist Gordon Gallup, Jr, attempts to determine an animal’s degree of self-recognition by placing a spot of odorless paint on the animal’s body (most often the face) while the animal is anesthetized. The test then entails simply observing the animal to see if it recognizes the spot using a mirror, and subsequently attempts to remove it. The test may seem rather rudimentary, but it actually requires a fairly high degree of cognitive complexity. The animal must not only recognize that a reflected image is oneself, but also must have a mental representation of what he or she normally looks like to recognize that the blemish is out of place. (Gallup Jr, Anderson, & Shillito, 2002)
So far the list of animals that have “passed” the mirror test is quite impressive. They include a wide variety of apes (1), elephants (2), pigs (3), dolphins (4), and another member of the corvid bird family, the magpie (5). ((1) Gallup Jr, 1970; (2) Plotnik, De Waal, & Reiss, 2006; (3) Broom, Sena, & Moynihan, 2009; (4) Reiss & Marino, 2001; (5) Prior, Schwarz, & Güntürkün, 2008)
The mirror test is somewhat lacking in scope because it assumes that an organism’s sensory repertoire is dominated by vision (more than 50% of the human brain participates in processing visual information). Many animals rely more on other senses to navigate the world. For example, dogs tend to rely most heavily on olfaction. But until we come up with a smell-o-vision mirror, we will be hard pressed to get Otis to pass the mirror test (although I have little doubt that he could).
Let’s move on from the recognition of one’s reflection to the recognition of one’s thoughts. Knowing that we know allows us as humans to critically evaluate our own conscious experience. This ability feels very human, but as is becoming a trend in this article, the ability extends beyond the human realm.
Rhesus monkeys are able to report the presence or absence of a memory in a testing environment. The monkeys were taught that touching one shape on a computer triggered a memory test that, if answered correctly, yielded a highly valued treat while another shape did not require a test but provided a much less valuable piece of food. The memory test consisted of a set of 4 symbols, one of which, when touched, would provide the high value treat while a wrong answer yielded no food. If the monkey were successful on the memory test, the triggering symbol wouldn’t be changed and the monkey could use its memory to select the correct symbol on a subsequent test.
The experiment hinged on varying the time delay between tests. If the monkeys had a delay of only 30 seconds they would choose to touch the shape triggering the memory test because they remembered which subsequent symbol out of the 4 memory test options would yield a high value treat. Yet when experimenters increased the delay to 4 minutes, the monkeys often chose to press the shape associated with a low value food item instead of risking a wrong answer on the memory test.
Experimenters were able to demonstrate that the monkeys were accurately able to decide whether or not their memories were sufficient to engage in the memory test or to settle for the low value sure thing. These results suggest that the monkeys were able to examine their own memories and cognitions to at least a low level of abstraction. (Hampton, 2001)

ANIMAL TOOL USE “A Bonobo at the San Diego Zoo “fishing” for termites“. Licensed under CC BY-SA 3.0 via Wikimedia Commons.
Okay, so animals have similar brains, are conscious of visual images, can remember and infer, recognize themselves in the mirror, and can think about their own thoughts. Is there anything that is uniquely human? Maybe the prototypical use of tools? Nope, animals use tools too!
Asian elephants will strip and modify a tree branch to use as a fly swatter (1), chimpanzees will fold a leaf into a pouch for storing water (2), and New Caledonian crows (another corvid) will use twigs as hooks to dig bugs out of unreachable crevices (3). ((1) Hart, Hart, McCoy, & Sarath, 2001; (2) Tonooka, 2001; (3) Hunt & Gray, 2003)
These seem like relatively basic uses of tools. Are there any more complex examples? Absolutely!
Chimpanzees in zoos who are taught to exchange tokens for food will begin to save the tokens, choosing to spend them at their leisure (Sousa & Matsuzawa, 2001). And if New Caledonian crows are given a piece of straight, but bendable wire, they will bend the end into a hook to facilitate their grub foraging (Weir, Chappell, & Kacelnik, 2002). The bendable wire has no natural analog that the bird could have had previous experience with, so the bending represents a novel manipulation of the mental hook construct.
Now that we have exhausted nearly every “uniquely” human component of consciousness, let’s turn to the use of language.
Although no animal has yet to be identified that uses language to the degree that humans do, there are plenty of examples to dispel the myth that humans have the corner on the language market. Language is a loaded term so we will instead briefly investigate animal communication.
Chimpanzees can be taught to communicate with both sign language and through adapted keyboards. Even more remarkable, these same chimpanzees will then use sign language to communicate with each other in the absence of humans, suggesting that they have integrated the representational sign language into their own consciousness. (Fouts, Jensvold, & Fouts, 2002)
The African grey parrot goes far beyond mimicking its trainer and can assemble novel and abstract word and symbol combinations to identify objects, express desires (hunger, etc.), and emotional states (frustration). (Pepperberg, 2007)

VERVET MONKEY “Vervet monkey Feb09” by Taken byfir0002 | flagstaffotos.com.auCanon 5D II + Canon 400mm f/5.6 L – Own work. Licensed under GFDL 1.2 via Wikimedia Commons.
Animals in their natural environment also use complex communication to exchange information. The vervet monkey has 3 distinct warning calls that correspond to 3 different types of predators. Importantly, the vervet’s calls are not stereotyped reactions to viewing the predator because they will sometimes withhold the call if the situation requires it. (Seyfarth, Cheney, & Marler, 1980)
Mongoose warning calls are not only specific to the predator type, but also the volume corresponds to the intensity of the danger (Manser, Seyfarth, & Cheney, 2002). And finally, prairie dog calls specific to particular predators can be recorded and played back for other prairie dogs, producing the same response (Kiriazis & Slobodchikoff, 2006). In other words, the prairie dog calls are standardized within the species.
This has been a brief tour of the various components of animal consciousness. We did not even delve into the study of classical conditioning, a form of non-reflexive (1) associative learning requiring some mental representation and awareness (2) that can be acquired by organisms ranging from the invertebrate honey bee (3) to the squirrel. ((1) Rescorla, 1988; (2) Lovibond & Shanks, 2002; (3) Bitterman, Menzel, Fietz, & Schäfer, 1983)
Hopefully, I have convinced you that the chance that animals experience consciousness is quite a bit closer to 99% than 50%. I must disclose that I personally believe that we can ascribe a 100% chance that animals possess at least the most rudimentary of conscious experiences, but again this is an opinion and I will allow the reader to make up his or her own mind regarding animal consciousness.
For the purposes of the remaining paragraphs, let’s imagine that the weight of experimental and observational evidence has convinced us, like the neuroscientists of the Cambridge Declaration, that animals do in fact experience consciousness. What are the implications of this conclusion?
If animals are conscious, then what can they teach us?
Again, we need to be careful not to anthropomorphize animal consciousness. When I ask what animals can teach us I do not mean that their cognition is easily translated into human consciousness, or vice versa. I am simply suggesting that if we accept that animals are conscious in their own species-specific way, what can we learn by treating them as non-mechanistic beings? I don’t want to argue that we should try to act like a dog or a cow, I simply wish to suggest that we might be able to pick and choose some of their behavioral approaches to the world as an alternative conscious interpretation of our shared environment.
The allegorical nature of the human mind allows us to view a dog’s ability to love unconditionally as an aspiratory principle for ourselves. We need not lick the face of everyone we come in contact with to extract meaning from the expansive heart of our favorite furry companions. Instead of literal interpretations, we can translate what it would mean in human terms to love unconditionally. The powerful lessons of the natural world are written in prose, not prescriptions.
To study what we can gather from the observation of animals I will cite Otis as an example both because he is the most proximal animal (he is currently chewing a bone at my feet) and because, as a dog, he represents the most popular (by household) species of pet in the United States.

NATURE “Nature reserve Žižkův vrch in summer 2012 (21)” by Chmee2 – Own work. Licensed under CC BY-SA 3.0 via Wikimedia Commons.
“Nature does not hurry, yet everything is accomplished.” – Tao Te Ching by Lao Tsu (Feng & English, 1972)
Taoism (pronounced dow-ism) is an ancient Chinese philosophy that emphasizes the harmonization of one’s life with the “Way,” nature, or natural order. Taoism emphasizes an effortless interaction with the realities of the world. A complete Taoist philosophy is far outside of the scope of this article, but a simplistic understanding will suffice for the ensuing discussion.
Nature only lends its creations life. The symmetry of death is contained within every birth. This is the natural order of things. People, careers, events, and relationships are born and will eventually die. Every beginning must have an ending, both literally and metaphorically.
The animal brain evolved to seek out reward, avoid pain, and struggle for survival – in other words, to seek life and avoid death. These biological guidelines for behavior were extremely adaptive for our early ancestors, and they led to the tremendous evolutionary success of the kingdom Animalia and, in particular, the class Mammalia.
And yet, as consciousness evolved it elaborated ever-greater complexity onto the simplistic 3-part reward-pain-survival triad of adaptive interaction with the environment. The logical conclusion of this elaboration process is exemplified in the brains of the subspecies Homo sapiens sapiens (Latin for “wise, wise man”), modern humans. For in the human brain, the 3-part behavioral imperative expanded from the confines of the ever-present Now to the unpredictable future and murky past. And with this expansion of timeframe came new psychic problems.
No longer did each moment provide the salve for the moment that came before or stood to follow it. In the expanse of human consciousness, the 3-part survival guide grew to include chapters for the Tomorrows and Yesterdays that perpetually fan out on either side of Today. Human consciousness became a victim of its own success, and anxiety, regret, and existential fear were born.
Because of our evolutionary neurobiology we are programmed to cling to rewards and abhor loss. And in our Tomorrow and Yesterday-dominated consciousness we desire only reward and unending happiness; anything less is a disappointment.
But in a world whose natural state is equal parts birth and death, our desires stand to be forever disappointed. We cannot have perpetual happiness, good health, and bliss. We need the contrasting agents of sadness, sickness, and death to provide substance for their counterpart elements.
So what does all of this have to do with Otis and the floral rug preamble? Well as it turns out, the wisdom of a more natural consciousness is embodied in the plants, animals, and natural elements that surround us. Little did I know, but my own Taoist master has been trying to dig his way into my mind to teach me about naturalness, action without intent (a Taoist concept known as wu-wei), and humility.
When Otis decided to go spelunking into the depths of the rug’s floral pattern, the first question that I asked myself was “Why?” But what if this were the wrong question to ask? What if there were no “Why?” What if Otis digs just to dig?
Dogs are unbelievably wise. Yes, they eat items of questionable nutritional merit and rage against harmless objects such as vacuums, but contained within this behavior is a naturalness that exemplifies Taoist philosophy.
Humans have great difficulty performing a task without a purposeful goal in mind, whether it is taking a nap or digging a hole. Dogs are not hampered by this rational conundrum. When dogs nap, they just nap. And when they dig, they just dig. There is no mental elaboration, reasoning, or qualm that keeps them from fully embodying whatever behavior they are performing.
As it turns out, purposeless action is the key that opens the door to the Now. This is not to say that we cannot achieve the goals, benefits, and pleasures that are the natural byproduct of this purposeless action. Instead, “purposeless” refers to the relaxed state of mind during said action. When we paint, write, or engage in other creative endeavors, we “purposelessly” flow through the action, lost in the process despite a goal being present at some level of consciousness. Tomorrows and Yesterdays will never disappear from the human mental arena, but the suffering we experience when we cling to temporality outside of the Now can be moderated.
Simplicity has its own advantages. Otis, and ostensibly other animals, experiences a consciousness that is relatively immune to human worry or regret because it lacks some of the convolution. And although this lack of complexity may decrease the ability to form highly complex emotional or mental experiences, it does not obviate all emotional and mental experience. There is every bit of evidence that animal consciousness is enriched with an approximation of the emotional states and social representations that we so cherish (Griffin & Speck, 2004). Animals show us that we can depart from our intellectual neuroticism without losing the profundity of conscious experience.
Otis’s life consists of a series of moments that are not overly compared to the past or future. When my wife and I leave for the hospital in the morning Otis sits by the door for 5-10 minutes (we have a camera we can watch him on – and, yes, we are those parents), hoping that we will return. But after about 5-10 minutes he either lies down right where he stood or returns to his bed and dreams of carpet swatches and fluffy shag rugs. And when either my wife or I come home at lunchtime to let Otis out, he greets us without indignation, joyfully trotting outside to do his business and hopefully partake in a gentlemanly game of Frisbee.
Because his consciousness is in accord with the natural order of things, Otis is free to occupy each moment equally and fully. Even the moments of sadness or grief (such as when we first leave for work) are taken in stride. For animal wisdom does not evade natural suffering, it simply does not elaborate on a given hardship.
The key to Otis’s success seems to be his fluidity. If his owners run into a colleague on a walk and carry on a stationary conversation for too long, Otis will simply lie down and rest his eyes. And when the walk resumes, Otis pops up and enthusiastically resumes his search for the next great smell or fluffy squirrel. When Otis gets sick and exorcises his breakfast all over the living room rug, he simply experiences the nausea for a few moments, and then begins to re-consume the now pureed deliciousness.
I could go on and on about Otis’s uninhibited depressurizations of his digestive tract, his love of lying across his owners legs to prevent our quick escape, or his profound fear of any and all inanimate objects, but I think that I have gotten my point across. Animal consciousness, once established, reveals the incredibly wise Taoist teachers that surround us everyday.
Next time you drive past a group of cows, pat your dog on its head, or clean out the litter box while your cat watches disparagingly, I hope that you will pause your own conscious stream long enough to consider the wisdom that dwells within animal consciousness.
References
Allen, Colin, Trestman, & Michael. (2014). Animal Consciousness. (Zalta ed.) The Stanford Encyclopedia of Philosophy. http://plato.stanford.edu/archives/sum2014/entries/consciousness-animal/. Retrieved 3/19/15.
Bitterman, M. E., Menzel, R., Fietz, A., & Schäfer, S. (1983). Classical conditioning of proboscis extension in honey bees (Apis mellifera). Journal of Comparative Psychology, 97(2), 107.
Broom, D. M., Sena, H., & Moynihan, K. L. (2009). Pigs learn what a mirror image represents and use it to obtain information. Animal Behaviour, 78(5), 1037-1041.
Clayton, N. S., & Dickinson, A. (1998). Episodic-like memory during cache recovery by scrub jays. Nature, 395(6699), 272-274.
Cavalli-Sforza, L. L., & Feldman, M. W. (2003). The application of molecular genetic approaches to the study of human evolution. nature genetics, 33, 266-275.
Darwin, C. (2009). The origin of species by means of natural selection: or, the preservation of favored races in the struggle for life. W. F. Bynum (Ed.). AL Burt.
Dickinson, A., & Balleine, B. (2002). The role of learning in the operation of motivational systems. Stevens’ handbook of experimental psychology.
Eccles, J. C. (1994). The evolution of consciousness. In How the SELF Controls Its BRAIN (pp. 113-124). Springer Berlin Heidelberg.
Edelman, G. M. (2003). Naturalizing consciousness: a theoretical framework. Proceedings of the National Academy of Sciences, 100(9), 5520-5524.
Edelman, G. M. (2004). Wider than the sky: The phenomenal gift of consciousness. Yale University Press.
Edelman, D. B., & Seth, A. K. (2009). Animal consciousness: a synthetic approach. Trends in neurosciences, 32(9), 476-484.
Emery, N. J., & Clayton, N. S. (2001). Effects of experience and social context on prospective caching strategies by scrub jays. Nature, 414(6862), 443-446.
Feng, G. F., & English, J. (1972). Lao Tsu: Tao Te Ching. Aldershot: Wildwood House.
Fouts, R. S., Jensvold, M. L. A., & Fouts, D. H. (2002). Chimpanzee signing: Darwinian realities and Cartesian delusions. The cognitive animal: empirical and theoretical perspectives on animal cognition. MIT Press, Cambridge, Mass, 285-291.
Gallup, G. G. (1970). Chimpanzees: self-recognition. Science, 167(3914), 86-87.
Gallup Jr, G. G., Anderson, J. R., & Shillito, D. J. (2002). The mirror test. The cognitive animal: Empirical and theoretical perspectives on animal cognition, 325-33.
Griffin, D. R. (2001). Animal minds: Beyond cognition to consciousness. University of Chicago Press.
Griffin, D. R., & Speck, G. B. (2004). New evidence of animal consciousness. Animal cognition, 7(1), 5-18.
Hallett, M. (2007). Volitional control of movement: the physiology of free will. Clinical Neurophysiology, 118(6), 1179-1192.
Hampton, R. R. (2001). Rhesus monkeys know when they remember. Proceedings of the National Academy of Sciences, 98(9), 5359-5362.
Harland, D. P., & Jackson, R. R. (2004). Portia perceptions: the Umwelt of an araneophagic jumping spider. Complex worlds from simpler nervous systems, 5-40.
Hart, B. L., Hart, L. A., McCoy, M., & Sarath, C. R. (2001). Cognitive behaviour in Asian elephants: use and modification of branches for fly switching. Animal Behaviour, 62(5), 839-847.
Hesslow, G. (2002). Conscious thought as simulation of behaviour and perception. Trends in cognitive sciences, 6(6), 242-247.
Hunt, G. R., & Gray, R. D. (2003). Diversification and cumulative evolution in New Caledonian crow tool manufacture. Proceedings of the Royal Society of London B: Biological Sciences, 270(1517), 867-874.
Kay, E. H., & Hoekstra, H. E. (2008). Rodents. Current Biology, 18(10), R406-R410.
Kiriazis, J., & Slobodchikoff, C. N. (2006). Perceptual specificity in the alarm calls of Gunnison’s prairie dogs. Behavioural Processes, 73(1), 29-35.
Lovibond, P. F., & Shanks, D. R. (2002). The role of awareness in Pavlovian conditioning: empirical evidence and theoretical implications. Journal of Experimental Psychology: Animal Behavior Processes, 28(1), 3.
Low, P., Panksepp, J., Reiss, D., Edelman, D., Van Swinderen, B., Low, P., & Koch, C. (2012). The Cambridge Declaration of Consciousness in Non-Human Animals.
Manser, M. B., Seyfarth, R. M., & Cheney, D. L. (2002). Suricate alarm calls signal predator class and urgency. Trends in cognitive sciences, 6(2), 55-57.
Nagel, T. (1974). What is it like to be a bat?. The philosophical review, 435-450.
Panksepp, J. (2005). Affective consciousness: Core emotional feelings in animals and humans. Consciousness and cognition, 14(1), 30-80.
Pepperberg, I. M. (2007). Grey parrots do not always ‘parrot’: the roles of imitation and phonological awareness in the creation of new labels from existing vocalizations. Language sciences, 29(1), 1-13.
Plotnik, J. M., De Waal, F. B., & Reiss, D. (2006). Self-recognition in an Asian elephant. Proceedings of the National Academy of Sciences, 103(45), 17053-17057.
Prior, H., Schwarz, A., & Güntürkün, O. (2008). Mirror-induced behavior in the magpie (Pica pica): evidence of self-recognition. PLoS biology, 6(8), e202.
Rees, G., Kreiman, G., & Koch, C. (2002). Neural correlates of consciousness in humans. Nature Reviews Neuroscience, 3(4), 261-270.
Reiss, D., & Marino, L. (2001). Mirror self-recognition in the bottlenose dolphin: A case of cognitive convergence. Proceedings of the National Academy of Sciences, 98(10), 5937-5942.
Rescorla, R. A. (1988). Pavlovian conditioning: It’s not what you think it is. American Psychologist, 43(3), 151.
Roth, G. (2000). The evolution and ontogeny of consciousness.
Seyfarth, R. M., Cheney, D. L., & Marler, P. (1980). Vervet monkey alarm calls: semantic communication in a free-ranging primate. Animal Behaviour, 28(4), 1070-1094.
Skinner, B. F. (1977). Why I am not a cognitive psychologist. Behaviorism, 1-10.
Sousa, C., & Matsuzawa, T. (2001). The use of tokens as rewards and tools by chimpanzees (Pan troglodytes). Animal Cognition, 4(3-4), 213-221.
Squire, L. R. (Ed.). (2013). Fundamental neuroscience. Academic Press.
Stoerig, P., Zontanou, A., & Cowey, A. (2002). Aware or unaware: assessment of cortical blindness in four men and a monkey. Cerebral Cortex, 12(6), 565-574.
Templer, V. L., & Hampton, R. R. (2013). Episodic memory in nonhuman animals. Current Biology, 23(17), R801-R806.
Tononi, G., & Edelman, G. M. (1998). Consciousness and complexity. science, 282(5395), 1846-1851.
Tonooka, R. (2001). Leaf-folding behavior for drinking water by wild chimpanzees (Pan troglodytes verus) at Bossou, Guinea. Animal cognition, 4(3-4), 325-334.
Ward, L. M. (2011). The thalamic dynamic core theory of conscious experience. Consciousness and cognition, 20(2), 464-486.
Watson, J. B. (1913). Psychology as the behaviorist views it. Psychological review, 20(2), 158.
Weir, A. A., Chappell, J., & Kacelnik, A. (2002). Shaping of hooks in New Caledonian crows. Science, 297(5583), 981-981.








![MRI by Nevit Dilmen / [CC-BY-SA-3.0 or GFDL]](/wp-content/uploads/2014/07/brain-color-e1406157479540-160x160.jpg)




I re-read this a few times. At many levels this is fascinating. I am looking at my four dogs and wonder if they know how lucky they are…..
Great information.
Thanks Patty! You have been a long time supporter of MindfulnessMD and the articles therein, I greatly appreciate your readership. I know animals and pets in particular are a subject close to both of our hearts so I’m glad that you had a chance to read this article. Thanks again!